| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website http://www.jofem.org |
Original Article
Volume 7, Number 6, December 2017, pages 172-177
The Impact of Vitamin D Status on Preoperative Ultrasound Findings and Parathyroidectomy Outcomes in Patients With Primary Hyperparathyroidism
Ashutosh Suryaa, Peter N. Taylorb, Hussam Abusahmina, Onyebuchi Okosiemea, Gautam Dasa, c
aDepartment of Diabetes and Endocrinology, Prince Charles Hospital, Cwm Taf University Health Board, Merthyr Tydfil, CF47 9DT, UK
bDepartment of Diabetes and Endocrinology, University Hospital of Wales, Cardiff and Vale University Health Board, Cardiff, CF14 4XW, UK
cCorresponding Author: Gautam Das, Department of Diabetes and Endocrinology, Prince Charles Hospital, Cwm Taf University Health Board, Merthyr Tydfil, CF47 9DT, UK
Manuscript submitted December 14, 2017, accepted December 20, 2017
Short title: Vitamin D, Ultrasound and Parathyroidectomy
doi: https://doi.org/10.14740/jem477w
| Abstract | ▴Top |
Background: An impaired vitamin D status may present with an aggressive phenotype of primary hyperparathyroidism (PHPT). We evaluated the impact of 25-hydroxyvitamin D (25(OH)D3) levels on pre-operative ultrasound findings and outcomes following parathyroidectomy.
Methods: Retrospective analysis of 81 patients who had parathyroidectomy for PHPT from January 2008 to October 2016. Patients were grouped according to their 25(OH)D3 levels (deficient (< 30 nmol/L), insufficient (≥ 30 and < 50 nmol/L), sufficient (≥ 50 nmol/L)). All patients had preoperative ultrasound scans (USS) and their pre-and post-operative biochemistry results and pathology reports were obtained from the hospital portal system.
Results: Preoperatively, there was no statistically significant difference in parathyroid hormone (PTH) (P = 0.19) and adjusted calcium (P = 0.91) levels in different vitamin D subgroups. Parathyroidectomy led to significant improvement of PTH, adjusted calcium, phosphate, alkaline phosphatase and creatinine levels (P < 0.00001). USS did not show any significant difference in localizing single gland disease (SGD) in vitamin D subgroups (sensitivity 57.1%, 62.5% and 55.8% in deficient, insufficient and sufficient groups respectively) and the chances of finding a negative scan was similar in all groups (about 44.5%). USS could not detect any multiple gland disease (MGD). Inferiorly located glands were more involved especially when 25(OH)D3 levels were in the deficient or insufficient range. Postoperatively, patients had higher adenoma weight in vitamin D deficient and insufficient groups (1.42 ± 1.35 g and 1.11 ± 1.39 g) compared to sufficient group (0.58 ± 0.48 g) for SGD and trend was identical for the combined weight of glands in MGD (1.81 ± 2.79 g; 0.93 ± 0.58 g and 0.59 ± 0.33 g respectively). Regression analysis showed an inverse association of 25(OH)D3 level with adenoma weight for SGD (r = -0.006; P = 0.008) but not when multiple glands were resected (r = -0.0007; P = 0.54).
Conclusions: Vitamin D does not influence biochemical severity of disease in PHPT. USS is a reliable tool for localization in SGD but ineffective for MGD and the sensitivity and specificity of scan results are not affected by vitamin D levels. Hypovitaminosis D leads to higher adenoma weight and an inverse relationship exists between the two for SGD.
Keywords: Vitamin D; Ultrasound; Primary hyperparathyroidism; Parathyroidectomy; Single gland disease; Multiple gland disease
| Introduction | ▴Top |
Vitamin D and parathormone (PTH) are two key regulators of calcium homeostasis and the interrelationship between these two hormones in patients with primary hyperparathyroidism (PHPT) has received considerable attention in the last 10 - 15 years. Low levels of vitamin D often overlap with PHPT and several studies have reported this association largely in the elderly patients [1-3]. Some observations suggest that in PHPT the conversion of 25-hydroxyvitamin D (25(OH)D3) to 24-hydroxylated component is increased thereby increasing the prevalence of vitamin D deficiency [4, 5] but other factors may also contribute to the co-existence of both these conditions [6]. It is not known whether vitamin D deficiency could trigger PHPT but suboptimal vitamin D status (insufficiency or deficiency) has been linked to more aggressive phenotype of PHPT with higher levels of PTH [7]. In addition, it may also lead to more pronounced parathyroid gland proliferation resulting in larger adenomas [8, 9] and higher markers of bone turnover with greater frequency of fractures [6].
Minimally invasive focussed parathyroid surgery is widely practised nowadays for PHPT and pre-operative parathyroid imaging have facilitated a paradigm shift in managing such patients. Imaging has limited ability to differentiate between etiologies of parathyroid pathology, but it facilitates improved localization, differentiates between single adenoma with multiple gland disease (MGD) leading to reduced operating time and maximizes operative cure. Amongst all available non-invasive techniques, ultrasonography and nuclear scintigraphy are the most commonly used modalities. Several studies have explored the correlation of pre-operative vitamin D status with predictive accuracy for localization and post-operative adenoma weight and biochemical parameters using the technetium (99 mTc) sestamibi scan in PHPT but the evidence of similar correlations using the ultrasound scan (USS) is very scarce and conflicting [10-14].
USS is inexpensive and widely available and has a sensitivity of 76-79% in identifying single adenoma’s which drops to 35% in patients with MGD [15, 16]. It can also make a correct prediction of laterality of single adenomas in 74% of patients with exact quadrant identification in 72% patients [17]. Few authors have published results of parathyroid localization utilizing USS in patients with PHPT with or without vitamin D deficiency but information on whether pre-operative vitamin D status influence anatomical characterization of these glands when visualized by USS is lacking. In this study we aimed to explore the role of vitamin D on USS findings done pre-operatively in patients with PHPT and investigated whether such findings have any relationship with post-operative biochemical parameters, parathyroid adenoma weight and histological outcomes.
| Materials and Methods | ▴Top |
Patients
This is a retrospective analysis of all patients who had a parathyroidectomy for PHPT in our centre from January 2008 till October 2016. The information was obtained from the computerized hospital medical records system utilizing appropriate coding for “primary hyperparathyroidism” and “parathyroidectomy”. We initially obtained data for 98 patients, but four patients were excluded due to wrong coding as they had parathyroidectomy for other indications. We also excluded patients who had insufficient or incomplete pre-or post-operative data (n = 13). Finally, 81 patients were enrolled for this study. The study was registered with the research and development division of Cwm Taf University health board (registration no: CT 813/17). We did not obtain any informed consent from patients for this study as we only accessed their data retrospectively from the hospital data base ensuring appropriate anonymity.
Biochemical measurements
Serum levels of total calcium, PTH (ref range: 1.6 - 6.9 pmol/L), phosphate (ref range: 0.8 - 1.5 mmol/L), alkaline phosphatase (ref range: 30 - 130 U/L) and creatinine (ref range: 46 - 92 µmol/L) were all analysed on Roche Modular P analysers (Roche, Burges Hill, UK). The adjusted calcium levels (ref range: 2.2 - 2.6 mmol/L) was calculated by an in house derived equation applied to the calcium to correct it to account for albumin concentration. The eGFR (ref range: > 90 ml/min/1.73 m2) was calculated using IDMS aligned MDRD equation: 175 × (((serum creatinine - 3.08)/1.004) × 0.011312)-1.154 × Age-0.203 × (0.742 if female). Serum TSH (ref range: 0.27 - 4.2 mU/L) and FT4 (ref range: 11 - 25 pmol/L) were determined by electro chemiluminescence on Roche Modular E analysers (Roche, Burges Hill, UK). 25(OH)D3 levels were performed by the Chromsystems MassChrom method (Munich, Germany) on an Agilent 6410 LC: MSMS (Cheadle, UK). USS was performed in the hospital radiology department using Phillips Medical systems IU22 and Epiq 5 (from May 2016 onwards only) machines. Post-operative information regarding macroscopic weight of parathyroid glands and histological findings were obtained from the pathology section of the hospital data reporting system (Welsh clinical portal). Patients were categorised into groups according to their 25(OH)D3 levels as vitamin D deficient (25(OH)D3 < 30 nmol/l); insufficient (25(OH)D3 ≥ 30 and < 50 nmol/L) and sufficient (25(OH)D3 ≥ 50 nmol/L).
Statistical analysis
All data are expressed as mean ± standard deviation (SD) and paired t test were used to compare the groups and one-way ANOVA to compare more than two groups. Logistic regression analysis was performed with 25(OH)D3 levels as a dependent variable and compared with weight of parathyroid gland in SGD and combined weight of glands in MGD. A P value of < 0.05 was regarded as statistically significant. All statistical analysis was performed using the statistical package SPSS for Windows (version 16.0, SPSS Inc, Chicago, IL, USA).
| Results | ▴Top |
Table 1 shows the baseline demographic and biochemical characteristics of patients at baseline and following parathyroidectomy (within 3 - 6 months). There was a female predominance (80.2%; n = 65/81) and majority of patients were elderly. Following surgery there was statistically significant lowering of mean levels of PTH, adjusted calcium and alkaline phosphatise (ALP) with improvement in phosphate and creatinine levels. Individually 78/81 (96.3%) patients achieved normocalcemia (adjusted calcium < 2.6 mmol/L) and 85.2% (69/81) patients had complete normalization of PTH levels. The average length of stay was about 4 days and the mean 25(OH)D3 level for the whole group demonstrated a high proportion of hypovitaminosis D.
 Click to view | Table 1. Patient Demographics and Biochemical Parameters Before and After Parathyroidectomy |
Table 2 demonstrates the USS findings are per 25(OH)D3 status. We observed that the likelihood of finding multiple gland involvement in PHPT by USS is negligible irrespective of vitamin D status and therefore, it may not be the best imaging modality for exclusion of MGD. Our data also shows that the chances of finding a negative scan (with no glandular enlargement) in different vitamin D subgroups were identical (about 44.5%). Interestingly, we also found that by USS criteria, the superiorly located glands (left and right) are minimally involved in PHPT but the left inferior gland is more involved when patients have hypovitaminosis D (deficient or insufficient) whereas the right inferior gland is more commonly enlarged when patients have their 25(OH)D3 levels in the sufficient range.
 Click to view | Table 2. Characteristics of Gland Involvement by Ultrasonography as per Vitamin D Status |
The overall sensitivity of the USS for parathyroid adenoma localization was 58.4% with a specificity of 100%. The sensitivity, specificity and positive predictive value of USS scans for adenoma localization was not significantly different in patients in low vitamin D group (insufficient and deficient) from the patients with normal vitamin D levels. The low negative predictive value in all categories indicates that USS may not the best modality for complete exclusion of adenomatous disease in PHPT when scans are negative (Table 3).
 Click to view | Table 3. Relationship Between Ultrasound Findings and Histology as per Vitamin D Status |
Table 4 shows that majority of our patients had single gland disease (n = 55) irrespective of vitamin D status but the chances of MGD and their removal were higher when patients had deficient (n = 15) or insufficient levels (n = 9) of vitamin D in comparison to patients whose vitamin D levels were optimal (n = 2). The weight of the glands in SGD and the combined weight of glands in MGD were higher in vitamin D deficient and insufficient patients when compared to the sufficient group but the difference across groups was not statistically significant. In both SGD and MGD the histology of majority of patients was suggestive of a benign adenoma in all vitamin D subgroups but chief cell adenomatous changes were principally encountered in the vitamin D deficient group and hyperplastic changes were noted mainly in patients with vitamin D insufficiency.
 Click to view | Table 4. Post Parathyroidectomy Findings According to Vitamin D Status |
We also performed a regression analysis with vitamin D as the dependent variable and established that an inverse correlation exists between 25(OH)D3 levels and the weight of a resected gland in SGD (r = -0.006; P = 0.008) (Fig. 1a) and such inverse relationship existed even after adjustment for age and sex (r = -0.009; P = 0.004). A similar relationship could not be established between 25(OH)D3 levels and the combined weight of resected glands in MGD (r = -0.0007; P = 0.54) (Fig. 1b).
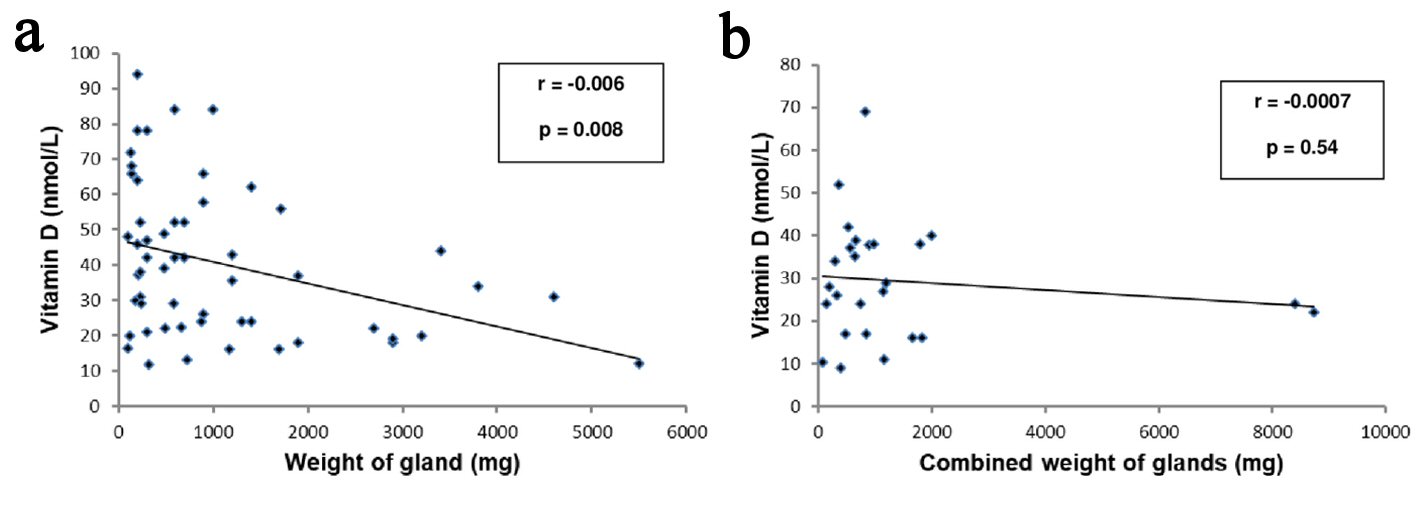 Click for large image | Figure 1. (a) Regression analysis with vitamin D as dependent variable with weight of parathyroid gland in patients with single gland disease. (b) Regression analysis with vitamin D as dependent variable with combined weight of parathyroid glands in patients with multiple gland disease. |
| Discussion | ▴Top |
PHPT remains a surgical disease and parathyroidectomy is beneficial in achieving normocalcemia and normal PTH levels along with improvement in other biochemical indices. The traditional approach for PHPT has been a four-gland exploration to define the extent of disease which needed less emphasis on preoperative imaging. In recent years, minimally invasive surgery has been more popular due to its smaller extent, shorter duration and lower rates of complication. This approach requires accurate pre-operative localization to direct surgery to the correct site. Parathyroid ultrasonography (US) and scintigraphy (SG) are the main methods used for this purpose and forms an integral part of most diagnostic algorithm. Although the complimentary role of US and SG has been reported in the literature [18] but none of them should be regarded as diagnostic as heterogeneous definitions are often used to evaluate effectiveness and in most cases of “negative” imaging, an experienced surgeon will locate the disease during exploration.
An altered vitamin D status is perhaps the most significant confounding factor found relating to patients with PHPT. Low levels of Vitamin D have been often linked to higher levels of PTH and larger size of parathyroid adenoma [9, 13], hence one would speculate that patients with impaired vitamin D status will have an aggressive phenotype of PHPT and the likelihood of pre-surgical localization would be higher due to chronic gland stimulation. In contradiction to this speculation, our study shows that presence of vitamin D insufficiency or deficiency does not affect the biochemical severity of disease at presentation. There is a greater predilection of the inferiorly located glands to be involved more with adenomatous changes in PHPT especially the right inferior gland which is mostly involved when patients have their vitamin D levels in the insufficient or deficient range. The reason for this remains speculative but could well be related to the peculiarity of blood supply to the inferiorly located glands.
Our findings also suggest that US is a reliable and effective tool for localizing SGD but in cases of negative localization where MGD is suspected proceeding to a SG would be the most reasonable option. While trying to establish a concurrence between US findings and histology outcomes we have established that US has a sensitivity of about 58% and specificity of 100% in identifying parathyroid adenoma which is compatible with other authors [14] but the findings do not overtly change when patients were subcategorised as per vitamin D status (sensitivity: 56-62%; specificity: 100%). We therefore conclude that although vitamin D deficiency may lead to an element of secondary hyperparathyroidism, it does not increase chances of pre-operative localization. Our findings do not concur with observations made by Kandil et al [12] who established that lower plasma levels of vitamin D were associated with positive radiological findings although the modality chosen for imaging was Tc99m sestamibi scan and not US.
We also noted a definite relationship between weight of the resected parathyroid gland and vitamin D status. As vitamin D deficiency is a stimulus for parathyroid hyperplasia and parathyroid tumour weight is the most reliable indicator of parathyroid cell mass, our patients who were vitamin D deficient or insufficient had the largest mean gland weight both in SGD and MGD and vitamin D sufficient patients had the smallest size glands. These findings echo with results from previous work by other authors [9, 13, 19]. We also noticed an inverse association between serum 25(OH)D3 levels and gland weight after resection especially in SGD which would be relevant in the management of PHPT as this would strongly add to the growing body of evidence whether vitamin D deficiency or insufficiency should be corrected routinely to reduce adenoma weight and disease severity before undertaking parathyroidectomy. We have also established that despite vitamin D deficiency or insufficiency patients are more likely to have SGD than MGD (48.1% (39/81) vs. 29.6% (24/81)). These findings are similar to Randle et al [7] who reported that MGD was less common in patients with severe vitamin D deficiency (19% vs. 30%, P = 0.06) and in a larger cohort of all vitamin D deficient patients (20% vs. 30%, P < 0.001). Although benign adenoma was the principal histological variant in our cohort of patients, we also established that chief cell adenoma was the second most common variant and is more prevalent in vitamin D deficiency state.
Our study is constrained by its retrospective design and the small sample size. Second, we did not include the findings of SG for preoperative definition or localization of glands as only 20% of our patients had a SG study. If this data was available for all patients our findings would have been more robust as SG and USS findings are often complimentary. Nevertheless, we have been able to establish that USS is an effective tool with comparative efficacy and accuracy to SG in pre-operative localization prior to parathyroid surgery when patients have variable vitamin D status. It will be a viable, low cost and low radiation option for centres that do not have the facility for SG. Third, our study does not explore the causal relationship of how vitamin D deficiency influence radiological and post-operative findings, but it paves the way for interventional prospective studies where pre-operative vitamin D replacement may be beneficial in patients with PHPT to reduce their disease burden.
In conclusion, our study adds to the growing evidence that vitamin D insufficiency or deficiency is a significant problem which co-exists with PHPT. An abnormal vitamin D status does not overtly impact biochemical severity of disease in PHPT. USS is a reliable tool for pre-operative localization of parathyroid glands, but imaging outcomes are not significantly underpinned on underlying vitamin D status. However, uncorrected vitamin D deficiency leads to higher adenoma weight and may potentially influence operative outcomes; hence, clinicians should consider replacing it routinely prior to parathyroidectomy.
Conflict of Interest
We do not have any conflict of interest.
Grant Support
We do not receive any support from any organization.
| References | ▴Top |
- Silverberg SJ. Vitamin D deficiency and primary hyperparathyroidism. J Bone Miner Res. 2007;22(Suppl 2):V100-104.
doi pubmed - Saliba W, Lavi I, Rennert HS, Rennert G. Vitamin D status in primary hyperparathyroidism. Eur J Intern Med. 2012;23(1):88-92.
doi pubmed - Yetley EA. Assessing the vitamin D status of the US population. Am J Clin Nutr. 2008;88(2):558S-564S.
pubmed - Souberbielle JC, Bienaime F, Cavalier E, Cormier C. Vitamin D and primary hyperparathyroidism (PHPT). Ann Endocrinol (Paris). 2012;73(3):165-169.
doi pubmed - Clements MR, Davies M, Hayes ME, Hickey CD, Lumb GA, Mawer EB, Adams PH. The role of 1,25 dihdroxyvitamin D in the mechanism of acquired vitamin D deficiency. Clin Endocinol (Oxf). 1992;37:17-27.
doi - Silverberg SJ, Shane E, Dempster DW, Bilezikian JP. The effects of vitamin D insufficiency in patients with primary hyperparathyroidism. Am J Med. 1999;107(6):561-567.
doi - Randle RW, Balentine CJ, Wendt E, Schneider DF, Chen H, Sippel RS. Should vitamin D deficiency be corrected before parathyroidectomy? J Surg Res. 2016;204(1):94-100.
doi pubmed - Grey A, Lucas J, Horne A, Gamble G, Davidson JS, Reid IR. Vitamin D repletion in patients with primary hyperparathyroidism and coexistent vitamin D insufficiency. J Clin Endocrinol Metab. 2005;90(4):2122-2126.
doi pubmed - Rao DS, Honasoge M, Divine GW, Phillips ER, Lee MW, Ansari MR, Talpos GB, et al. Effect of vitamin D nutrition on parathyroid adenoma weight: pathogenetic and clinical implications. J Clin Endocrinol Metab. 2000;85(3):1054-1058.
doi - Batawii N, Algithmi A. The effects of vitamin D level on the results of Tc99 sestamibi scans for parathyroid adenoma localization in patients with primary hyperparathyroidism. Int J Endocrinol Metab Disorder. 2017;3(2).
doi - Kim E, Song YS, Wong F, Lee DS, Perrier N. Correlation of 99 mTc sestamibi uptake with size and weight of parathyroid adenoma as well as serum levels of calcium, PTH and vitamin D. J Nucl Med. 2010;51:1565.
- Kandil E, Tufaro AP, Carson KA, Lin F, Somervell H, Farrag T, Dackiw A, et al. Correlation of plasma 25-hydroxyvitamin D levels with severity of primary hyperparathyroidism and likelihood of parathyroid adenoma localization on sestamibi scan. Arch Otolaryngol Head Neck Surg. 2008;134(10):1071-1075.
doi pubmed - Beyer TD, Chen EL, Nilubol N, Prinz RA, Solorzano CC. Short term outcomes of parathyroidectomy in patients with or without 25-hydroxy vitamin D insufficiency. Journal of Surgical Research. 2007;143:145-150.
doi pubmed - Tassone F, Castellano E, Gianotti L, Acchiardi F, Emmolo I, Attanasio R, Borretta G. Vitamin D deficiency does not affect the likelihood of pre-surgical localization in asymptomatic primary hyperparathyroidism. Endocrine Practice. 2016;22:205-209.
doi pubmed - Cheung K, Wang TS, Farrokhyar F, Roman SA, Sosa JA. A meta-analysis of preoperative localization techniques for patients with primary hyperparathyroidism. Ann Surg Oncol. 2012;19(2):577-583.
doi pubmed - Ruda JM, Hollenbeak CS, Stack BC, Jr. A systematic review of the diagnosis and treatment of primary hyperparathyroidism from 1995 to 2003. Otolaryngol Head Neck Surg. 2005;132(3):359-372.
doi pubmed - Siperstein A, Berber E, Mackey R, Alghoul M, Wagner K, Milas M. Prospective evaluation of sestamibi scan, ultrasonography, and rapid PTH to predict the success of limited exploration for sporadic primary hyperparathyroidism. Surgery. 2004;136(4):872-880.
doi pubmed - Hindie E, Ugur O, Fuster D, O’Doherty M, Grassetto G, Urena P, Kettle A, et al. 2009 EANM parathyroid guidelines. Eur J Nucl Med Mol Imaging. 2009;36(7):1201-1216.
doi pubmed - Untch BR, Barfield ME, Dar M, Dixit D, Leight GS, Jr., Olson JA, Jr. Impact of 25-hydroxyvitamin D deficiency on perioperative parathyroid hormone kinetics and results in patients with primary hyperparathyroidism. Surgery. 2007;142(6):1022-1026.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.









