| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website http://www.jofem.org |
Original Article
Volume 1, Number 1, April 2011, pages 27-37
Association of Younger Age With Poor Glycemic and Cholesterol Control in Asians With Type 2 Diabetes Mellitus in Singapore
Matthias Paul Han Sim Toha, c, Christine Xia Wua, Helen Soh Sum Leongb
aHealth Services and Outcomes Research, National Healthcare Group, Singapore
bClinical Services, National Healthcare Group Polyclinics, Singapore
cCorresponding author: Matthias Toh, Health Services and Outcomes Research, National Healthcare Group, 6 Commonwealth Lane, #04-01/02 GMTI Building, Singapore 149547
Manuscript accepted for publication April 7, 2011
Short title: Age and Type 2 Diabetes Control
doi: https://doi.org/10.4021/jem13e
| Abstract | ▴Top |
Background: The prevalence of type 2 diabetes mellitus (T2DM) in children and adolescents is increasing in both poor and rich countries. Epidemiologic studies have reported significant and continuous associations between HbA1c level and diabetes-related vascular complications. Younger adults with early exposure to hyperglycemia are at high risk for end-organ damage. Few studies have reported the relationship of age to glycemic control in patients with T2DM world-wide and little comparison among young adults and elderly patients especially in the Asian population. This study investigates the relationship of age and glycemic control in Asian patients with T2DM attending primary care clinics in Singapore.
Methods: We included T2DM patients who had at least 2 visits to the public-sector primary care clinic for diabetes treatment in 2009. Demographic characteristics, medical records and laboratory results were extracted from the enterprise-wide chronic disease registry. The mean HbA1c, blood pressure and LDL-cholesterol were trended by age. Multivariate logistic regression was used to identify the factors predicting “poor” glycemic control.
Results: There were 58,057 T2DM patients and both the mean HbA1c and LDL-cholesterol were lower among elderly than adult patients. Mean HbA1c was 8.08 ± 1.62% for patients < 45 years old and 6.86 ± 0.99% for patients 85+ years old. Mean LDL-cholesterol levels were 2.84 ± 0.81 and 2.55 ± 0.73 mmol/L for the respective age groups. The Malay and Indian groups had significantly poorer glycemic control compared to the Chinese, AdjOR 1.65 (95% CI: 1.54 - 1.77) and 1.50 (95% CI: 1.40 - 1.61) respectively. Other significant predictors of poor glycemic control included the male gender, presence of maculopathy or retinopathy, peripheral vascular disease, coronary heart disease, heart failure, and being on insulin therapy (AdjOR 8.00; 95% CI: 7.54 - 8.48). Patients with poor LDL-c (4.1+ mmol/L) were 4.2 times more likely to have poor glycemic control (95% CI: 3.78 - 4.66) while those with Grade 2 hypertension were 1.5 times (95% CI: 1.35 - 1.76) more likely than those without hypertension.
Conclusions: Younger T2DM patients had poorer glycemic and cholesterol control than older patients in Singapore and they should have targeted interventions to achieve “optimal” glycemic and cholesterol control.
Keywords: Glycemia; LDL-cholesterol; Control; Younger; Type 2 diabetes mellitus
| Introduction | ▴Top |
In Singapore, the prevalence of diabetes mellitus increased from 8.2% in 2004 to 11.5% in 2010 amongst its population aged between 18 and 69 years [1]. The increase in diabetes prevalence was observed across all ages, in both genders and all the major ethnic groups, especially the Malays. The prevalence estimates for 2010 (age-standardised to World Standard Population) from the International Diabetes Federation [2] indicate that Singapore’s prevalence is much higher than other Asian countries such as Hong Kong (8.5%), Taiwan (7.5%), South Korea (5.3%) and Japan (5.0%), and other Western countries such as France (6.7%), Australia (5.7%) and the United Kingdom (3.6%).
Type 2 diabetes mellitus (T2DM) is no longer a disease of middle aged and older individuals [3]. Its prevalence in children and adolescents is on the increase in all countries, whether poor or rich [2]. Epidemiologic and prospective studies have reported significant and continuous associations between HbA1c level and diabetes-related vascular complications [4-9]. Younger adults with early exposure to hyperglycemia are at high risk for end-organ damage [4-8]. As with type 1 diabetes, many children with T2DM risk developing complications at an early age, adding to a significant burden on the family and society. There is growing recognition that T2DM in the young is fast becoming a global public health issue with a potentially serious health outcome [10].
Comprehensive and appropriate management of patients with diabetes should include early screening for complications and optimize control of glucose, blood pressure and cholesterol. Very few studies have reported the relationship of age to glycemic control in patients with T2DM and the focus was largely on middle-aged and elderly patients [11]. Conclusions have been mixed, showing high prevalence of poor control in elderly, better glycemic control in older patients [12], or no effect of age on metabolic control [13]. In 2003, El-Kebbi et al studied the relationship of age to glycemic control in an African American population and showed a high prevalence of obesity and poor glycemic control in young adults compared to older patients [14]. A German study in 2009 also reported that the highest percentage of patients with inadequate glycemic control in the T2DM population is not found among the old but the 45 - 54 and 55 - 64 age groups [15].
To date, there has been little comparison of the glycemic control in young adults and elderly patients especially in the Asian population. The aim of this current study was to investigate the relationship of age to glycemic control in Asian patients with T2DM attending the public-sector primary care clinics in Singapore and identify the factors predicting poor glycemic control.
| Materials and Methods | ▴Top |
This is a retrospective study of patients attending the National Healthcare Group Polyclinics (NHGP) for the treatment of diabetes mellitus in 2009. The NHGP is a chain of 9 public sector primary care clinics providing basic comprehensive care for the population in the central and western parts of Singapore. It has an integrated electronic patient medical record which hosts both administrative and clinical information. Medical records of all patients with chronic disease conditions such as diabetes mellitus, hypertension, dyslipidemia, stroke, and coronary heart disease are linked to the enterprise-wide National Healthcare Group (NHG) Disease Management System (CDMS) [16].
Study population
We selected all the patients with existing diagnosis of T2DM from the NHG CDMS who had at least 2 attendances in the same clinic in 2009. Patients who were newly diagnosed with type 2 diabetes mellitus in 2009 or those with type 1 diabetes mellitus were excluded from the study.
Study parameters and data collection
All the demographic characteristics, medical diagnosis, clinical parameters and laboratory results were extracted directly from the NHG CDMS. Demographic characteristics included age, gender and ethnic group. Medical conditions were extracted based on ICD-9 CM diagnosis codes for hypertension, dyslipidemia, coronary heart disease, stroke, retinopathy, and peripheral vascular disease. Body Mass Index (BMI) and Glomerular Filtration Rate (GFR) were extracted and classified according to WHO guidelines.
Glycated hemoglobin (HbA1c) was the marker for measuring glycemic control. The HbA1c results in 2009 were obtained and a mean HbA1c was calculated for every patient. Glycemic control was considered “optimal” if HbA1c was ≤ 7.0%, “acceptable” if HbA1c was 7.1 - 8.0%, and “poor” if HbA1c was above 8.0%. Age was grouped into bands of 10 years for comparison of glycemic control.
Two other intermediate outcome measures were also profiled with age bands. The latest blood pressure (both systolic, SBP, and diastolic blood pressure, DBP) and LDL-cholesterol (LDL-c) levels in 2009 were recorded. BP control was considered “optimal” if SBP was ≤ 130 mmHg and DBP was ≤ 80 mmHg, “acceptable” if SBP was 131 - 139 mmHg and DBP was 81 - 89 mmHg, and “poor” if SBP was ≥ 140 mmHg and DBP was ≥ 90 mmHg. Dyslipidemia control was considered “optimal” if LDL-c was < 2.6 mmol/L, “acceptable” if LDL-c was 2.6 - 3.3 mmol/L and “poor” if LDL-c was ≥ 3.4 mmol/L.
Data analysis
Data was analyzed using PASW (version 18.0). Significance testing of proportions was carried out using Chi-square test, and of means using analysis of variance (ANOVA), where a probability (P) of less than 0.05 was considered significant. Multivariate logistic regression was used to study the factors predicting “poor” glycemic control.
This study was approved by the NHG Domain Specific Review Board.
| Results | ▴Top |
There were 58,057 patients with T2DM from 9 primary care clinics in the study. Table 1 shows that females outnumbered males by 54% : 46%. There were more males than females in the younger age groups up to 54 years and the reverse was observed from 55 years and above. Age is normally distributed (mean 64.0 ± 11.6 years), with 64% aged 60 years and older. Overall, the disproportionately higher proportion of Indians (13%) in relation to the general Singapore population reflected the higher incidence of diabetes mellitus in this ethnic group. The Indians and Malays made up 23% and 19% respectively among those below 45 years old, declining to below 10% for patients 75 years and above.
 Click to view | Table 1. Characteristics of Patients With Type 2 Diabetes Mellitus in National Healthcare Group Polyclinics in 2009 |
Other medical conditions
The prevalence of hypertension and dyslipidemia among these T2DM patients were 84% and 98% respectively. The prevalence of hypertension increased with age from 53% (below 45 years) to above 90% (65 years and above). The prevalence of dyslipidemia was high for all age groups, ranging from 95% to 99%.
The prevalence of vascular disease conditions increased with age. About 1 in 4 patients were also treated for coronary heart disease (range 5% to 40%), 11% had history of a cerebrovascular event (range 2% to 26%), 9% had retinopathy (range 6% to 10%) and 4% had peripheral vascular disease (2% to 8%). The proportion of patients with Chronic Kidney Disease (CKD) stages 1 and 2 decreased with age (Table 1).
BMI distribution
Mean BMI was 26.3 ± 4.7 kg/m2. There were more patients being overweight or obese among the younger age groups compared to older age groups (Table 1). About 1% to 4% was underweight, with increasing prevalence among the old age groups. A larger proportion of the older patients, especially those 85 years and older, did not have a weight measurement within the year.
Glycemic control
Mean HbA1c decreased with age, from 8.08 ± 1.62% for those below 45 years to less than 7% for those 85 years and above (Table 2). The distribution of HbA1c across the age groups is shown in Figure 1. The proportion of patients in each age group with HbA1c > 8% reduced with age, from 40% (below 45 years) to 10% (85 years and above).
 Click to view | Table 2. Mean HbA1c, LDL-c, Serum Creatinine and Blood Pressure by Age Group |
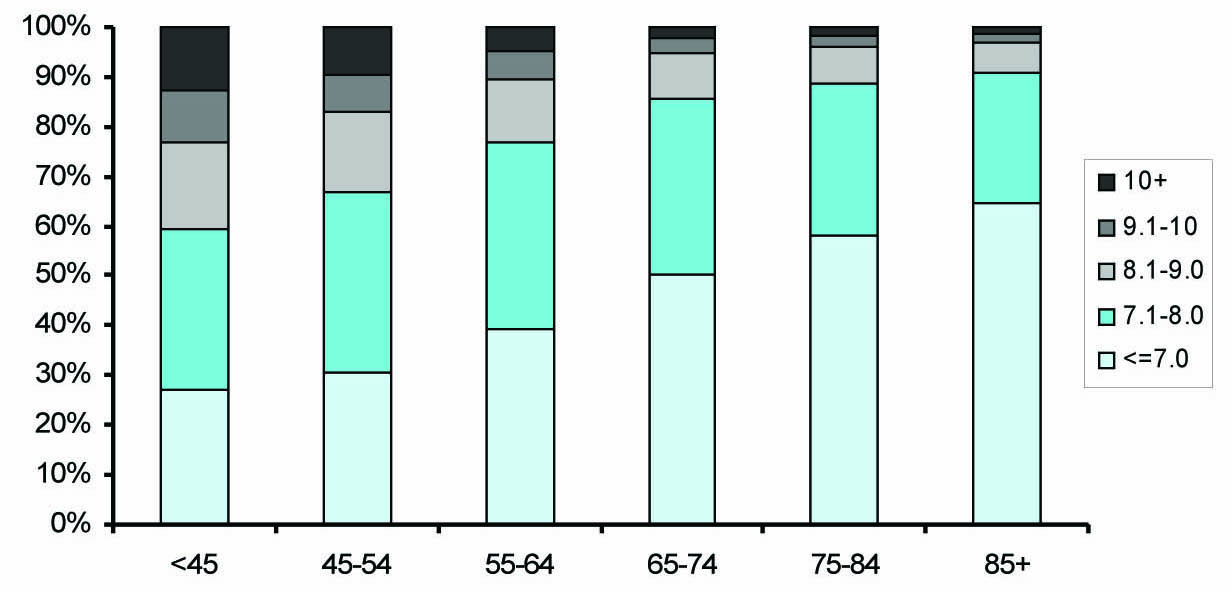 Click for large image | Figure 1. Distribution of mean HbA1c (%) by age group. |
Blood pressure control
The mean blood pressure was 131/73 mmHg (Table 2). When comparing across the age groups, the proportion with “normal” and “high normal” blood pressure control reduced with age from 71.2% (below 45 years) to 55% (85 years and older).
LDL-cholesterol (LDL-c) control
Similar to HbA1c, the mean LDL-c decreased with age, from 2.84 ± 0.81 mmol/L (below 45 years) to 2.55 ± 0.73 mmol/L (85 years and older). Proportion with “optimal” LDL-c control increased from 42% (below 45 years) to 57% (85 years and above) (Fig. 2).
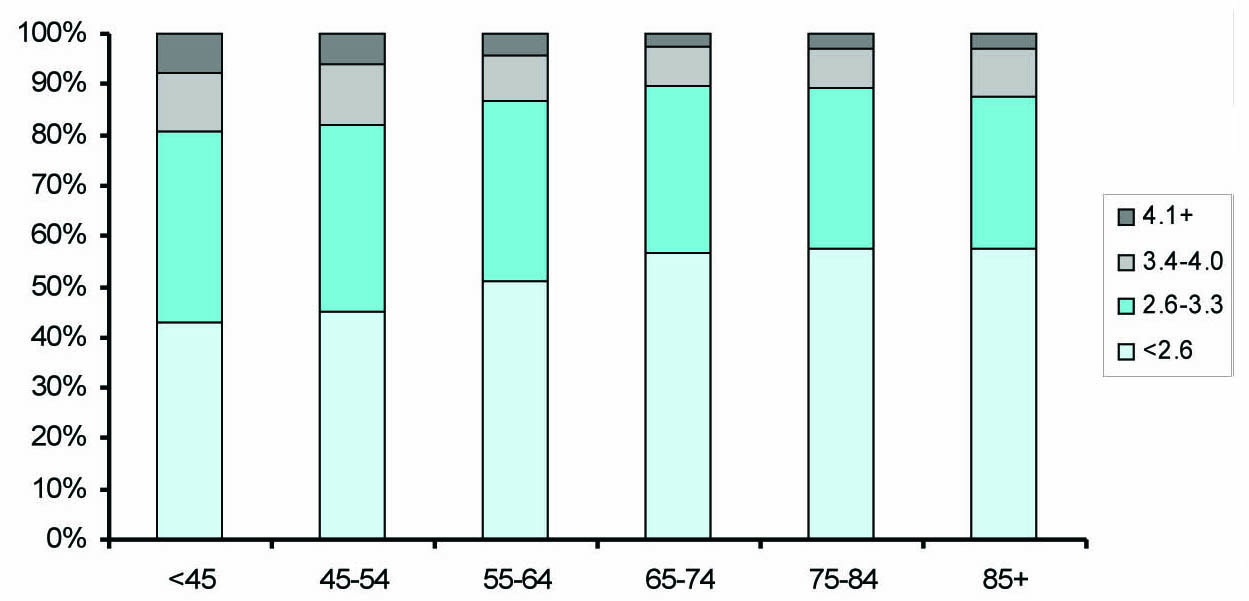 Click for large image | Figure 2. Distribution of mean LDL-cholesterol (mmol/L) by age group. |
Management of diabetes mellitus
The average number of physician clinic visits per year for diabetes care increased with HbA1c level, from 4.1 (for patients with HbA1c below 7%) to 5.2 (for patients with HbA1c 9 - 10%) and 4.9 (for patients with HbA1c above 10%). The proportion of patients who visited a Care Manager at least once in the year also increased with HbA1c, from 6.8% (HbA1c below 7%) to 52% (HbA1c above 9%). Similarly, percentage of patients with visits to the Dietitian increased from 1.3% (HbA1c below 7%) to 10% (HbA1c above 9%).
The proportion of patients in each age group treated with insulin ranged from 6.2% to 15.4% and was associated with the degree of glycemic control, increasing from 30.3% (for patients with mean HbA1c 8.1 to 9.0%) to 45.5% (for patients with mean HbA1c above 10%).
Multivariate logistic regression analysis
Table 3 shows the results of the multivariate logistic regression to predict “poor” HbA1c level above 8%. Compared to the group 85 years and above, those below 75 years old were significantly more likely to have “poor” HbA1c control. The adjusted OR for < 45 years was the highest (4.21; 95% CI: 3.38 - 5.26), followed by 45 - 54 years (3.24; 95% CI: 2.64 - 3.97), 55 - 64 years (2.12; 95% CI: 1.74 - 2.59) and 65 - 74 years (1.31; 95% CI: 1.07 - 1.59). Male patients had an adjusted OR 1.21 (95% CI: 1.15 - 1.27) over the female patients. The Malay and Indian groups had significantly poorer glycemic control compared to the Chinese, with adjusted OR of 1.65 (95% CI: 1.54 - 1.77) and 1.50 (95% CI: 1.40 - 1.61) respectively.
 Click to view | Table 3. Logistic Regression for Factors Significantly* Associated With “Poor” Glycemic Control |
Patients with known cardiovascular complications had higher odds of “poor” glycemic control than those without maculopathy or retinopathy, peripheral vascular disease, coronary heart disease or heart failure (Table 3). Patients who were treated with insulin were 8.19 (95% CI: 7.70 - 8.73) times the odds for having “poor” glycemic control than those on oral hypoglycemic agents only. Those with “poor” glycemic control also had correspondingly poorer LDL-c and blood pressure control. Compared to patients with “optimal” LDL-c < 2.6 mmol/L, the adjusted OR for “poor” glycemic control increased with poorer LDL-c control, from 1.42 (1.34 - 1.50) for group with LDL-c 2.6 - 3.3 mmol/L to 4.30 (3.87 - 4.78) for the group with LDL-c above 4.1 mmol/L. Similarly, compared to those with normal blood pressure, adjusted OR for “poor” glycemic control among grade 1 hypertension was 1.20 (1.12 - 1.70) and grade 2 hypertension was 1.47 (1.27 - 1.70).
| Discussion | ▴Top |
A few studies described the relationship of age to glycemic control in patients with diabetes [12, 13, 17]. In the Strong Heart Study among native Americans, age was found to be inversely related to HbA1c level [12]. There was no change in the median HbA1c level at baseline (1989 - 1992) and follow-up (1994 - 1995) surveys. The NHANES III (1988 - 1994) also reported that younger patients were more likely to have an elevated HbA1c level, although there was no significant association between age and HbA1c levels in a predominantly white population [13]. In an Australian study, Bruce et al also reported that age was inversely associated with glycemic control whereas duration of diabetes and treatment with either oral hypoglycemic agents or insulin were positively associated with glycemic control [18]. It was noted that octogenarians in Australia differed significantly from younger age groups, those with longer diabetes duration did not demonstrate the increase in hyperglycemia seen in other age groups. A significantly greater proportion of the oldest diabetic subjects had satisfactory HbA1c levels compared with younger subjects.
The Diabcare-Asia project from Singapore, India and Taiwan had earlier reported that one-third to one-half of the diabetic population had poor glycemic control and suboptimal lipid control [19-21]. Our study also supports that, in a predominantly Asian patient population attending primary care clinics, the younger patients had poorer HbA1c and LDL-c control than older patients. The prevalence of hypertension and dyslipidemia were high across all ages and higher than the prevalence in the general population. As expected, the prevalence of vascular complications such as coronary heart disease and cerebrovascular disease increased with age and were higher than the general population without diabetes mellitus [22-25].
Studies have suggested that early onset T2DM was associated with an increased risk for complications compared with later onset diabetes [26] and that the development and progression of complications might be more rapid in early onset disease [27-29]. Song et al in 2009 reported that the management of risk factors for diabetes complications was inadequate among the early onset T2DM cohort and they were at substantial risk of developing diabetes complications in later years and at an earlier stage [30]. Our study also showed that the poor glycemic control among the younger T2DM patients was associated with poorer cholesterol and blood pressure control. These younger patients have a higher lifetime risk of developing micro- and macro-vascular complications and should be treated much more aggressively to achieve “optimal” glycemic, blood pressure and cholesterol control [7, 9, 22, 31-36].
It is not fully understood why younger patients have worse glycemic control than older patients. In Singapore, all citizens regardless of age have equal access to medical care provided by the public sector. The older patients may be more motivated to take care of their diabetes and are more compliant with their medication and eat healthy low-fat diet [37]. On the other hand, younger patients might be more likely to disregard diabetes as being important and be less adherent to medication, lifestyle and diet restrictions. Similar findings were reported by El-Kebbi in 2003 that the persistence of HbA1c elevation in younger individuals could be due to inadequately low medication dosage or infrequent use of combination drug regimens [14]. The younger patient also tends to be more obese than older patients with resultant higher insulin resistance and may need more aggressive therapy to achieve glycemic control. In addition to treating raised HbA1c, physicians should be alerted to commence or reinforce aggressive lifestyle intervention, lipid-lowering and anti-hypertensive therapy especially for the younger T2DM patients.
This study also showed that patients with “optimal” glycemic control had fewer visits to the clinic annually for the treatment of diabetes. Those with “poor” glycemic control were more likely to have visited a Care Manager and Dietitian for general self-management tips, health information and dietary advice on diabetes. The overall healthcare utilization and expenditure would be correspondingly higher for patients with poorer glycemic control. Wagner showed thata sustained reduction in HbA1c level among adult diabetic patients is associated with significant cost savings within 1 to 2 years of improvement [38].
There are several limitations in this study. As data was drawn from the Diabetes Registry, we were unable to collect data to adjust for the duration of diabetes mellitus which may be associated with progressive impairment of insulin secretion. There was also no data on physical activity and adherence of diet and lifestyle. Some patients, especially the older ones, did not have BMI measurement.
Nonetheless, this study of an Asian population with diabetes mellitus has provided an insight into the variation of glycemic and cholesterol control with age. We analyzed the data of a large captive population from the Diabetes Registry and we were able to study the prevalence of hypertension, dyslipidemia and other vascular complications in patients with T2DM. All laboratory results were captured directly and were accurate and complete. The mean HbA1c for the year was calculated for every patient to reflect the average glycemic control over a year instead of using a single HbA1c reading.
Younger diabetics are at higher cumulative risk to develop vascular-related complications over time. The reasons for poor glycemic and cholesterol control are not well understood and are likely to be multifactoral. Future research could study patient’s health literacy and their understanding of diabetes, health-seeking and treatment-adherence behavior across the ages.
This study shows that younger patients with T2DM had poorer glycemic and cholesterol control than older patients in Singapore. Those with poor glycemic control also had corresponding poorer cholesterol and blood pressure control. These patients had a higher lifetime risk of developing micro- and macro-vascular complications and more research should be done to investigate reasons for the poorer control so that targeted interventions can be designed for them.
Acknowledgments
We would like to thank the patients and clinicians at NHG Polyclinics for contributing data for this study and members of the NHG Diabetes Disease Management Workgroup for their contribution to the CDMS and study. We would also like to thank Mr. Fong Chee Weng from theEpidemiology & Disease Control Division of Ministry of Health in Singapore for providing information on the National Health Survey results.
Financial Disclosure
The authors declare that they have no relevant financial interests in this manuscript.
| References | ▴Top |
- www.channelnewsasia.com/stories/singaporelocalnews/view/1095709/1/.html of 26 Nov 2010 1632 hrs.
- Diabetes Atlas Committee. Diabetes Atlas. 3rd ed. Brussels (Belgium): International Diabetes Federation, 2006.
- Zimmet PZ, McCarty DJ, de Courten MP. The global epidemiology of non-insulin-dependent diabetes mellitus and the metabolic syndrome. J Diabetes Complications 1997;11(2):60-68.
pubmed doi - Klein R. Hyperglycemia and microvascular and macrovascular disease in diabetes. Diabetes Care 1995;18(2):258-268.
pubmed doi - Kuusisto J, Mykkanen L, Pyorala K, Laakso M. NIDDM and its metabolic control predict coronary heart disease in elderly subjects. Diabetes 1994;43(8):960-967.
pubmed doi - The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993;329(14):977-986.
pubmed - Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;352(9131):837-853.
pubmed - Ohkubo Y, Kishikawa H, Araki E, Miyata T, Isami S, Motoyoshi S, Kojima Y, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract 1995;28(2):103-117.
pubmed doi - Elley CR, Kenealy T, Robinson E, Drury PL. Glycated haemoglobin and cardiovascular outcomes in people with Type 2 diabetes: a large prospective cohort study. Diabet Med 2008;25(11):1295-1301.
pubmed - Fagot-Campagna A, Narayan KM, Imperatore G. Type 2 diabetes in children. BMJ 2001;322(7283):377-378.
pubmed doi - Smith NL, Heckbert SR, Bittner VA, Savage PJ, Barzilay JI, Dobs AS, Psaty BM. Antidiabetic treatment trends in a cohort of elderly people with diabetes. The cardiovascular health study, 1989-1997. Diabetes Care 1999;22(5):736-742.
pubmed doi - Hu D, Henderson JA, Welty TK, Lee ET, Jablonski KA, Magee MF, Robbins DC, et al. Glycemic control in diabetic American Indians. Longitudinal data from the Strong Heart Study. Diabetes Care 1999;22(11):1802-1807.
pubmed doi - Shorr RI, Franse LV, Resnick HE, Di Bari M, Johnson KC, Pahor M. Glycemic control of older adults with type 2 diabetes: findings from the Third National Health and Nutrition Examination Survey, 1988-1994. J Am Geriatr Soc 2000;48(3):264-267.
pubmed - El-Kebbi IM, Cook CB, Ziemer DC, Miller CD, Gallina DL, Phillips LS. Association of younger age with poor glycemic control and obesity in urban african americans with type 2 diabetes. Arch Intern Med 2003;163(1):69-75.
pubmed doi - Huppertz E, Pieper L, Klotsche J, Stridde E, Pittrow D, Bohler S, Lehnert H. Diabetes Mellitus in German Primary Care: quality of glycaemic control and subpopulations not well controlled - results of the DETECT study. Exp Clin Endocrinol Diabetes 2009;117(1):6-14.
pubmed doi - Toh MP, Leong HS, Lim BK. Development of a diabetes registry to improve quality of care in the National Healthcare Group in Singapore. Ann Acad Med Singapore 2009;38(6):546-551.
pubmed - Carter JS, Gilliland SS, Perez GE, Skipper B, Gilliland FD. Public health and clinical implications of high hemoglobin A1c levels and weight in younger adult Native American people with diabetes. Arch Intern Med 2000;160(22):3471-3476.
pubmed doi - Bruce DG, Davis WA, Davis TM. Glycemic control in older subjects with type 2 diabetes mellitus in the Fremantle Diabetes Study. J Am Geriatr Soc 2000;48(11):1449-1453.
pubmed - Lee WR, Lim HS, Thai AC, Chew WL, Emmanuel S, Goh LG, Lau HC, et al. A window on the current status of diabetes mellitus in Singapore—the Diabcare-Singapore 1998 study. Singapore Med J 2001;42(11):501-507.
pubmed - Raheja BS, Kapur A, Bhoraskar A, Sathe SR, Jorgensen LN, Moorthi SR, Pendsey S, et al. DiabCare Asia—India Study: diabetes care in India—current status. J Assoc Physicians India 2001;49:717-722.
pubmed - Chuang LM, Tsai ST, Huang BY, Tai TY. The status of diabetes control in Asia—a cross-sectional survey of 24 317 patients with diabetes mellitus in 1998. Diabet Med 2002;19(12):978-985.
pubmed doi - Preis SR, Pencina MJ, Hwang SJ, D'Agostino RB, Sr., Savage PJ, Levy D, Fox CS. Trends in cardiovascular disease risk factors in individuals with and without diabetes mellitus in the Framingham Heart Study. Circulation 2009;120(3):212-220.
pubmed doi - Fox CS, Coady S, Sorlie PD, Levy D, Meigs JB, D'Agostino RB, Sr., Wilson PW, et al. Trends in cardiovascular complications of diabetes. JAMA 2004;292(20):2495-2499.
pubmed doi - Fox CS, Coady S, Sorlie PD, D'Agostino RB, Sr., Pencina MJ, Vasan RS, Meigs JB, et al. Increasing cardiovascular disease burden due to diabetes mellitus: the Framingham Heart Study. Circulation 2007;115(12):1544-1550.
pubmed doi - Selvin E, Coresh J, Golden SH, Brancati FL, Folsom AR, Steffes MW. Glycemic control and coronary heart disease risk in persons with and without diabetes: the atherosclerosis risk in communities study. Arch Intern Med 2005;165(16):1910-1916.
pubmed doi - Hillier TA, Pedula KL. Complications in young adults with early-onset type 2 diabetes: losing the relative protection of youth. Diabetes Care 2003;26(11):2999-3005.
pubmed doi - Pinhas-Hamiel O, Zeitler P. Acute and chronic complications of type 2 diabetes mellitus in children and adolescents. Lancet 2007;369(9575):1823-1831.
pubmed doi - Pavkov ME, Bennett PH, Knowler WC, Krakoff J, Sievers ML, Nelson RG. Effect of youth-onset type 2 diabetes mellitus on incidence of end-stage renal disease and mortality in young and middle-aged Pima Indians. JAMA 2006;296(4):421-426.
pubmed doi - Chuang LM, Soegondo S, Soewondo P, Young-Seol K, Mohamed M, Dalisay E, Go R, et al. Comparisons of the outcomes on control, type of management and complications status in early onset and late onset type 2 diabetes in Asia. Diabetes Res Clin Pract 2006;71(2):146-155.
pubmed doi - Song SH, Hardisty CA. Early onset type 2 diabetes mellitus: a harbinger for complications in later years—clinical observation from a secondary care cohort. QJM 2009;102(11):799-806.
pubmed - Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998;317(7160):703-713.
pubmed - Shepherd J, Barter P, Carmena R, Deedwania P, Fruchart JC, Haffner S, Hsia J, et al. Effect of lowering LDL cholesterol substantially below currently recommended levels in patients with coronary heart disease and diabetes: the Treating to New Targets (TNT) study. Diabetes Care 2006;29(6):1220-1226.
pubmed doi - Hansson L, Zanchetti A, Carruthers SG, Dahlof B, Elmfeldt D, Julius S, Menard J, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet 1998;351(9118):1755-1762.
pubmed doi - Collins R, Armitage J, Parish S, Sleigh P, Peto R. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Lancet 2003;361(9374):2005-2016.
pubmed - Gerstein HC, Miller ME, Byington RP, Goff DC, Jr., Bigger JT, Buse JB, Cushman WC, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358(24):2545-2559.
pubmed - Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, Marre M, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358(24):2560-2572.
pubmed - Glasgow RE, Hampson SE, Strycker LA, Ruggiero L. Personal-model beliefs and social-environmental barriers related to diabetes self-management. Diabetes Care 1997;20(4):556-561.
pubmed doi - Wagner EH, Sandhu N, Newton KM, McCulloch DK, Ramsey SD, Grothaus LC. Effect of improved glycemic control on health care costs and utilization. JAMA 2001;285(2):182-189.
pubmed doi
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.









