| Journal of Endocrinology and Metabolism, ISSN 1923-2861 print, 1923-287X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Endocrinol Metab and Elmer Press Inc |
| Journal website https://www.jofem.org |
Original Article
Volume 11, Number 5, October 2021, pages 108-114
Total Calcium and Ionized Calcium (pH = 7.4) Tests Are Equal When Screening Patients for Hypercalcemia at Admission in the Emergency Department: A Retrospective Descriptive Study
Regitze Wittenberga, c, Trine Rennebod Larsenb, Lis Stilgrena, Steen Antonsenb
aDepartment of Internal Medicine and Acute Medicine, Odense University Hospital, Svendborg Hospital, Valdemarsgade 53, 5700 Svendborg, Denmark
bDepartment of Biochemistry, Odense University Hospital, Svendborg Hospital, Valdemarsgade 53, 5700 Svendborg, Denmark
cCorresponding Author: Regitze Wittenberg, Department of Internal Medicine and Acute Medicine, Odense University Hospital, Svendborg Hospital, Valdemarsgade 53, 5700 Svendborg, Denmark
Manuscript submitted July 15, 2021, accepted August 2, 2021, published online October 31, 2021
Short title: Screening for Hypercalcemia in the ED
doi: https://doi.org/10.14740/jem757
| Abstract | ▴Top |
Background: Ionized calcium (pH = 7.4) (CaI) is a laborious test compared to tests for total calcium (CaT). However, CaI is the biologically active part of calcium in the blood and is therefore often considered the most relevant measure of calcium status. In this study, CaI and CaT tests were compared by identifying clinically relevant hypercalcemia (HC) in unselected patients acutely admitted to an emergency department (ED).
Methods: CaI and CaT were measured in all medical patients admitted to the ED at a single Danish Hospital (N = 13,400) between January 2018 and May 2019. The majority (97%) of the patients were admitted 1 - 3 times summing up to a total of 17,838 admissions. As limits for clinically relevant HC, 1.45 mmol/L for CaI and 2.77 mmol/L for CaT were used. Mismatches were defined as one test being equal to or above the limit while the other test was within or below its reference interval. In cases of mismatch, the medical record was reviewed.
Results: Seventeen mismatches with CaI ≥ 1.45 mmol/L were observed in 16 patients, of whom eight were known with diseases with risk of HC. Five patients had HC with no clinical relevance, one had HC of unknown relevance, and only two patients were discovered as having a prior unknown calcium metabolic disease. Three mismatches were observed in admissions with CaT ≥ 2.77 mmol/L. Mismatches did in none of these cases have any clinical consequences.
Conclusion: By using CaT as a screening method for HC instead of CaI, only two patients with prior unrecognized HC would have been missed and it did not result in a large number of patients with false HC. We therefore find that CaT is an acceptable test when screening for HC in the ED.
Keywords: Ionized calcium; Total calcium; Hypercalcemia
| Introduction | ▴Top |
Calcium is the most abundant mineral in the human body. The vast majority of the calcium (> 99%) is stored in bones while a minor fraction is located in the blood where it is found in three fractions. Approximately 40% is bound to plasma proteins, primarily albumin, approximately 10% is bound to anions, and the remaining half exists as ionized calcium (CaI). Calcium in blood is measured as the total amount of “bound and ionized” calcium (CaT) or as CaI [1, 2].
As CaI is the biologically active part of calcium in the blood, it is often considered the most relevant measure of calcium. However, multiple factors affect both tests. As a significant part of calcium is bound to plasma proteins, changes in these have major influence on CaT but only minor influence on CaI. CaI, on the other hand, is dependent on changes in pH and is corrected when measured in venous serum to the theoretical concentration at pH = 7.4. Furthermore, tests for CaI are difficult to automate and thus are laborious compared to tests for CaT, and in many parts of the world, CaI is not available in large numbers in routine clinical settings [3, 4].
The most typical causes for hypercalcemia (HC) are malignancy, primary hyperparathyroidism (PHPT), tertiary hyperparathyroidism, sarcoidosis or vitamin D poisoning [5, 6]. Symptoms of HC can be nonspecific, and therefore acutely admitted patients are often screened for HC. However, it is not clear whether CaI or CaT should be used for screening in this clinical setting.
The aim of this study was to compare tests for CaI and CaT in terms of identifying patients with previously unrecognized and clinically relevant HC among unselected medical patients being acutely admitted to an emergency department (ED). To our knowledge, there are no other studies that have looked into this clinical aspect.
| Materials and Methods | ▴Top |
This was a retrospective descriptive study performed at the ED at Odense University Hospital, Svendborg Hospital. According to the policies of the Ethical Committees of the Region of Southern Denmark, this project was exempt from ethics review. However, permission to access selected patient records was granted by the executive board of the hospital, as prescribed by local policies.
Patients
Our study population consisted of 13,400 patients (age 63.9 ± 19.9 years), 6,871 women (51.3%; age 63.9 ± 21.0 years) and 6,529 men (48.7%; age 64.0 ± 18.7 years). The patients had a total of 17,838 admissions in the study period. Most patients, 97% (13,024/13,400), were admitted one, two or three times, and only a minor fraction of 0.09% (12/13,400) was admitted more than 10 times. Mean age and standard deviation (SD) were calculated from the age of the patients at their first admission in cases of more than one admission during the study period.
Biochemical data
Prior to this study, CaI was used in the ED at Svendborg Hospital when screening for HC, but in the study period from January 2018 to May 2019, both months included, both CaI and CaT were included in the requesting profile for all acutely admitted medical patients. From the laboratory information system (LIS), BCC (CGI, Denmark), all requests from the ED from the period containing both CaI, CaT and plasma albumin (P-Alb) were extracted and made pseudonymous. Data for the individual patients were divided into admissions, defined as clusters of blood samples separated by intervals of minimum 7 days without blood drawings. Only the first blood sample from each admission, containing all three parameters, was included in the study.
CaI (pH = 7.4) in serum was measured on a Nova8 CRT (Novabiomedicals, Waltham, MA), while CaT and P-Alb in heparinized plasma were measured on a Cobas8000 (Roche Diagnostics GmbH, Mannheim, Germany).
Results of CaI (reference interval: 1.18 - 1.32 mmol/L) and CaT (reference interval: 2.15 - 2.51 mmol/L) were grouped according to the reference intervals of both tests. Elevated results were further divided if they were under or above a concentration of 1.45 mmol/L for CaI and 2.77 mmol/L for CaT. Both limits were chosen according to an international consensus report concerning patients with PHPT [7]. The report suggests, that surgery is indicated, when calcium levels are > 0.12 mmol/L above the upper limit for CaI and > 0.25 mmol/L above the upper limit for CaT. Mismatches of CaI and CaT were defined as one test being ≥ 1.45 mmol/L or ≥ 2.77 mmol/L, respectively, while the other test was within its reference interval or lower. Cases where CaI or CaT levels were above the upper reference limits, but below the mismatch limits, while the other test was within or below the reference interval, were defined as discrepancies.
For albumin, age-dependent reference intervals of 36 - 50 g/L (18 - 40 years), 36 - 48 g/L (40-70 years), and 34 - 45 g/L (> 70 years) were used. Albumin-corrected calcium (CaC) was measured by using correction formulas from Thode et al [8] and BMJ77 [9].
Patient records
In case of mismatch, the patient’s medical records were systematically reviewed and searched for the following: 1) the cause of the admission; 2) current and prior comorbidity; and 3) additional biochemistry. Comorbidity of interest was primarily diseases that could affect the calcium metabolism such as calcium metabolic diseases, chronic kidney disease (CKD) and cancer, including multiple myeloma. For patients with known calcium metabolic disease, it was noted if the patient was followed in an endocrinological outpatient clinic.
| Results | ▴Top |
During the observed period, a total of 17,838 admissions to the ED with patients > 18 years old were registered (Fig. 1). CaI was within the reference range in 13,928 (78.1%) admissions and under the reference range in 1,683 (9.4%) admissions. In the remaining 2,227 admissions (12.5%), HC was present (Table 1).
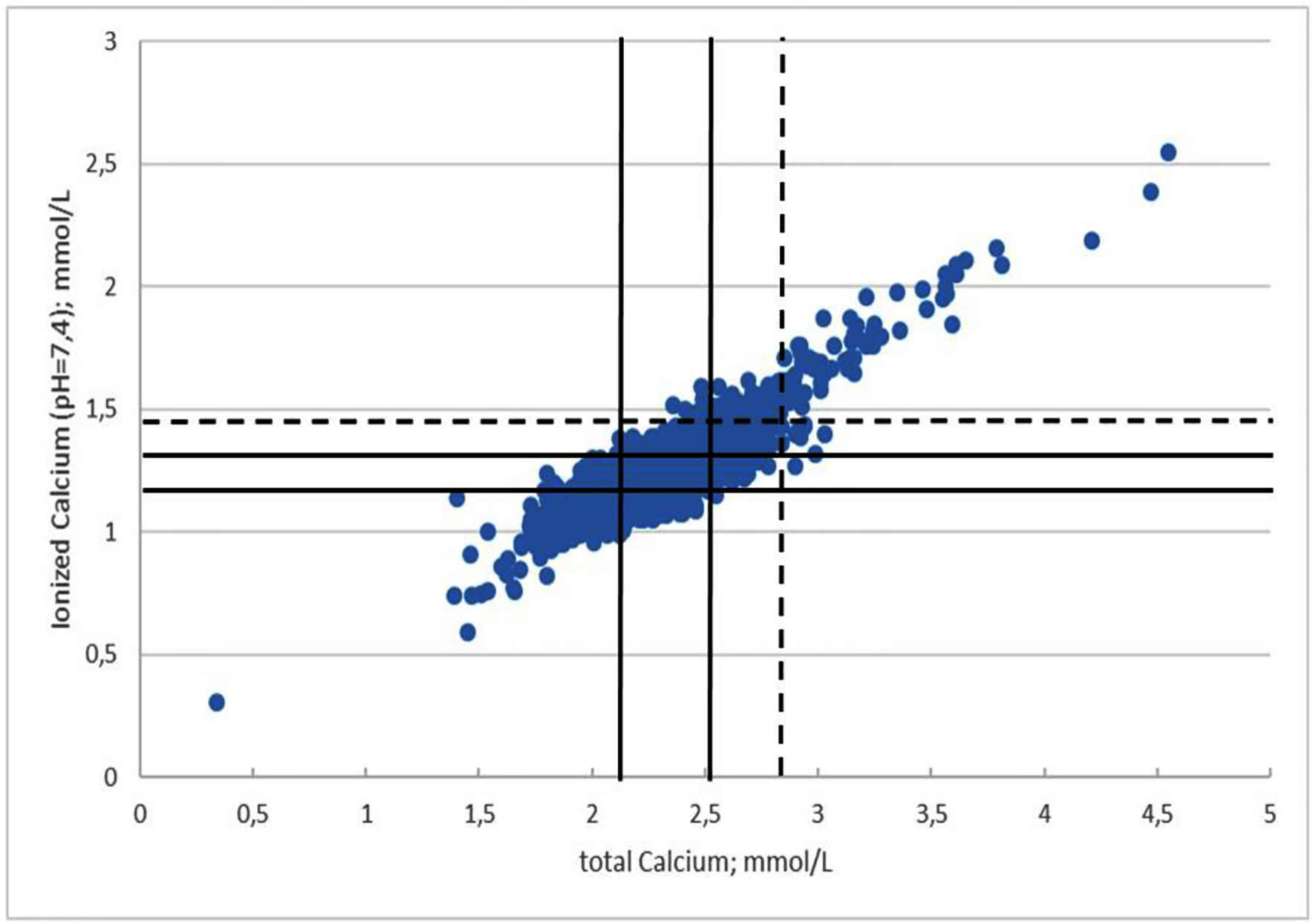 Click for large image | Figure 1. Distribution of CaI and CaT in 17,838 admissions. Unbroken lines represent reference limits for CaT and CaI. Dotted lines represent the limits for hypercalcemia. CaT: total calcium; CaI: ionized calcium (pH = 7.4). |
 Click to view | Table 1. Admissions According to Levels of CaT and CaI in mmol/L |
In 1,491 of the HC admissions, we observed an elevated CaI between 1.33 and 1.44 mmol/L combined with a value of CaT within or under the reference range giving a total discrepancy rate of 67% (1,491/2,227). In 17 admissions, CaI was above 1.44 mmol/L combined with a normal CaT, giving a mismatch rate of 0.8% (17/2,227).
Correspondingly, CaT was elevated in 962 admissions. In 25% (240/962) of these admissions, discrepancy was found, while mismatch was seen in 0.3% (3/962) admissions.
Patients with CaI mismatches
The 17 admissions with mismatch at CaI ≥ 1.45 mmol/L consisted of 16 different patients (age 73.4 ± 15.4 years), of whom one patient had two admissions within the same month. Thirteen (81%) of the patients were women. The highest measured CaI was 1.59 mmol/L (Table 2).
 Click to view | Table 2. Mismatches |
The admissions courses were different and not pathognomonic for HC. The most frequently reported diagnoses were pneumonia (four patients) and impaired general condition (four patients). Other reasons for admissions were urine retention, fall, abdominal/chest/back pains, paraesthesia in hands, fever, erysipelas, and urine tract infection. One patient presented with appendicitis.
Overall, eight patients were already known with diseases in which HC can be seen. Five patients had HC with no clinical relevance, one patient had HC of uncertain relevance, and in two patients a calcium metabolic disease was discovered.
Calcium metabolic disease
Six of the patients had calcium metabolic diseases being either PHPT or familial hypocalciuric HC (FHH). Four of them were already diagnosed prior to their admission. One patient got the diagnosis after the admission and despite showing limited symptoms, the patient was offered parathyroidectomy (PTX) as he only had one kidney. The other patient was not further investigated but it was believed the patient had PHPT due to repeated measurements of HC in combination with elevated PTH.
Cancer
Four patients had cancer, of which three of them were already known before admission; one had multiple myeloma in terminal phase, one had breast cancer with bone metastasis, and one patient with cancer in the gastroesophageal junction (GEJ) was admitted because of severe back pain, and bone metastasis were found during the admission. In none of the cases mentioned above did the mismatch make a difference for the patients’ prognosis. The fourth patient underwent a surgery for appendicitis but afterwards pathology showed a neuroendocrine tumor (NET), grade 1, with tumor mass radically removed and the following computed tomography (CT) scan was without metastasis.
Kidney disease
Two patients were known with CKD before the admission, which can explain the HC, including the patient mentioned above with multiple myeloma.
Dehydration
Two patients’ HC were explained with a combination of dehydration and calcium supplement, and calcium normalized after a pause of medication and rehydration.
Unknown
Four patients, including the patient with an NET in appendix, had HC with no further explanation. In two cases, calcium normalized under the admission respectively from 1.48 to 1.32 mmol/L and 1.49 to 1.27 mmol/L, only treated with hydration. In one case CaI was measured to 1.48 mmol/L at the start of the admission and 2 h later 1.35 mmol/L in an arterial blood gas (pH = 7.46). No controls were done in the patient with NET, so it is uncertain if the patient had a calcium metabolic disease.
Patients with CaT mismatches
Three patients had CaT ≥ 2.77 mmol/L combined with a normal CaI. One patient was admitted with massive dehydration, which normalized following rehydration. Calcium was not controlled. One patient was admitted with perforated ulcer, sepsis, and an arterial blood gas showed pH = 7.27 with a corresponding CaI level at 1.88 mmol/L. This normalized under admission due to a normalization of pH. This patient was a well-known outpatient because of HC with no obvious reason. The last patient was known with terminal CKD and was admitted due to inguinal pain. The patient’s calcium was not controlled. Common for all patients were chronic diseases (liver cirrhosis, CKD, diabetes, pancreatitis), in some cases combined with impaired kidney function and an albumin level high in the reference interval. In none of the cases did the mismatches have any clinical consequences as none of the patients needed treatment for HC.
Albumin-correction
By using the correction formula from Thode et al (Supplementary Table 1, www.jofem.org), we found four CaI mismatches and 13 CaC mismatches. Overall CaC was elevated above the reference level in 1,613 admissions. The BMJ77 correction (Supplementary Table 2, www.jofem.org) resulted in 10 CaI mismatches and three CaC mismatches. In these corrections CaC was elevated in 951 admissions.
| Discussion | ▴Top |
A low relative number of mismatch patients of clinical importance was found in this study. Only 1‰ (17/17,838) of the unselected admissions demonstrated a mismatch with CaI ≥ 1.45 mmol/L, and these mismatches constituted 0.8% (17/2,227) of all admissions with HC. Furthermore, the majority of these patients were previously diagnosed with diseases with problems in the calcium metabolism and risk of HC. Only two patients were discovered as having previously unrecognized PHPT, one was diagnosed with bone metastasis, without any change in prognosis, and one was incidentally diagnosed with an NET in which case the HC was not further investigated. This shows that not only a few numbers of mismatches were found, but also that it had minimal clinical relevance not to use CaI to screen these patients for HC.
CaT as a screening test did not result in a large number of patients with false clinically important HC, as only three patients with CaT ≥ 2.77 mmol/L had a normal CaI.
Limits for HC were chosen in accordance with the international consensus report about PHPT, because treatment for HC is typically necessary at this level. Our main focus was the clinical aspect and, therefore, this limit was considered relevant. It is uncertain whether the limits are relevant when screening for HC due to cancer and bone metastasis and there may be a risk of not identifying some patients with malignant HC. However, in these cases we would expect some clinical symptoms pointing towards malignant diseases together with the increased result of the calcium test.
In accordance with our results, prior studies have also shown good correlations between CaI and CaT [10, 11]. These included a sensitivity above 0.90 in cases with critically high CaI (≥ 1.58 mmol/L) assessed in 12,118 analyses from 4,699 patients in an intensive care unit (ICU) and a substantial agreement between CaI and CaT with 82% of the patients classified correctly by using CaT from an assessment of 20,003 tests. In our study, the highest measured mismatch value was 1.59 mmol/L, meaning no critically high calcium value was missed by using CaT.
Albumin-corrected calcium
The two different correction formulas for CaC both resulted in fewer cases with CaI mismatches than CaT. However, we do find this gain inconsiderate, because the number of mismatches with CaT is already low and as shown in our study of marginal clinical relevance. CaT as a screening identified 962 admissions with hypercalcemia which should be followed up with a CaI control measurement. By using albumin correction as suggested by Thode et al we found 1,613 admissions with hypercalcemia and by using the correction suggested in BMJ77 951. If using Thode et al it would result in a larger amount of CaI control tests compared to CaT. BMJ77 is almost identical to CaT. In our study CaT was found to be a valid screening for calcium levels in the blood, and we do not find that the correction formulas will be a better choice. Unadjusted CaT has also been shown to be superior to different CaCs in studies with 6,549 and 5,055 patients, respectively [12, 13]. In these, the diagnostic accuracy of CaC was compared to CaT by looking at multiple, from literature known, adjustment formulas, and also by looking at local formulas especially constructed for a specific set of data. Undoubtedly, albumin affects the level of agreement between CaI and CaT, but it is challenging to find a CaC, which has proven to be a better marker for HC than CaT. It is possible that certain CaC might be better than CaT in specific clinical situations. However, this might lead to situations with a number of different CaCs to be used within the same hospital for different clinical problems. This would require strict attentiveness when the individual CaC should be used and might introduce a risk for misunderstanding and faults.
Hypocalcemia
In 9.4% (1,683/17,838) of the admissions hypocalcemia was found, and among these there were also cases with CaT within the reference range. Hypocalcemia is a common issue in acute illness and it can be difficult to treat, as correction of hypocalcemia in these patients first of all is a treatment of the underlying disease [2]. Therefore, these patients were not included in this study.
When to use CaI
CaI should be considered in a number of clinical situations such as calcium metabolic diseases [8, 14], CKD [15], low albumin, and cancer [16] including multiple myeloma [17].
CaI has been shown to be a more sensitive indicator of PHPT in cases of calcium metabolic disease and more linearly associated with adenoma size in a study with 269 patients who had all undergone PTX because of PHPT [14]. However, the study did find a correlation between CaI and CaT overall. In cases of suspicion of calcium metabolic disease, the use of CaI has likewise been recommended, but it has also been suggested, in accordance with our results, that CaT could be used for screening in general [8].
Patients with CKD may have altered concentrations of CO2 and albumin in the blood, which can affect the relationship between CaI and CaT, and a sensitivity of only 21.4% was found for diagnosis of HC using CaT versus CaI in a study with 691 patients with CKD. Therefore, it was concluded, that CaI should be preferred in these patients [15].
Cancer diseases also have a known risk for HC. However, it has been found that patients with solid tumors and mildly increased calcium usually did not develop frank HC at follow-up and that the HC did not seem to influence on the prognosis. Therefore, it was suggested, that CaI should not be measured routinely in these patients [16]. Yet, in cases of multiple myeloma, which affects the bones, we think that CaI should be preferred [17]. However, as shown in this study CaT is sufficient for identifying HC in many clinical situations, and because CaT is much easier to produce in the lab compared to CaI, CaT should be chosen when possible and relevant.
Strengths and limitations
The strengths of this study include the size of the study both in terms of patients and admissions and that inclusion was consequent with every acutely admitted medical patient receiving a standard set of blood tests performed at hospitalization resulting in both CaI and Cat being measured at every admission. A limitation is the retrospective design of the study which means that one patient was classified as having HC of uncertain reason because no further investigations were made.
In conclusion, we found CaT to be an acceptable test when patients are screened for clinically relevant HC, in the setting of acutely admitted medical patients in an ED.
| Supplementary Material | ▴Top |
Suppl 1. Admissions According to Levels of Albumin-Corrected Calcium (CaC, Thode) and Ionized Calcium (pH = 7.4) in mmol/L
Suppl 2. Admissions According to Levels of Albumin-Corrected Calcium (CaC, BMJ77) and Ionized Calcium (pH = 7.4) in mmol/L
Acknowledgments
The extraction of data from LIS by technician Leif Pennerup is appreciated.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Content
Not applicable.
Author Contributions
SA and LS designed the study; SA and TRL performed data analyses; RW and LS reviewed patient records; RW wrote the manuscript. All authors reviewed, discussed and accepted the final version of the paper.
Data Availability
Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
| References | ▴Top |
- Peacock M. Calcium metabolism in health and disease. Clin J Am Soc Nephrol. 2010;5(Suppl 1):S23-30.
doi pubmed - Baird GS. Ionized calcium. Clin Chim Acta. 2011;412(9-10):696-701.
doi pubmed - Robertson WG, Marshall RW. Ionized calcium in body fluids. Crit Rev Clin Lab Sci. 1981;15(2):85-125.
doi pubmed - Larsen TR, Galthen-Sorensen M, Antonsen S. Ionized calcium measurements are influenced by albumin—should ionized calcium be corrected? Scand J Clin Lab Invest. 2014;74(6):515-523.
doi pubmed - Mirrakhimov AE. Hypercalcemia of malignancy: an update on pathogenesis and management. N Am J Med Sci. 2015;7(11):483-493.
doi pubmed - Turner JJO. Hypercalcaemia - presentation and management. Clin Med (Lond). 2017;17(3):270-273.
doi pubmed - Khan AA, Hanley DA, Rizzoli R, Bollerslev J, Young JE, Rejnmark L, Thakker R, et al. Primary hyperparathyroidism: review and recommendations on evaluation, diagnosis, and management. A Canadian and international consensus. Osteoporos Int. 2017;28(1):1-19.
doi pubmed - Thode J, Juul-Jorgensen B, Bhatia HM, Kjaerulf-Nielsen M, Bartels PD, Fogh-Andersen N, Siggaard-Andersen O. Comparison of serum total calcium, albumin-corrected total calcium, and ionized calcium in 1213 patients with suspected calcium disorders. Scand J Clin Lab Invest. 1989;49(3):217-223.
pubmed - Correcting the calcium. Br Med J. 1977;1(6061):598.
pubmed - Hu ZD, Huang YL, Wang MY, Hu GJ, Han YQ. Predictive accuracy of serum total calcium for both critically high and critically low ionized calcium in critical illness. J Clin Lab Anal. 2018;32(9):e22589.
doi pubmed - Ridefelt P, Helmersson-Karlqvist J. Albumin adjustment of total calcium does not improve the estimation of calcium status. Scand J Clin Lab Invest. 2017;77(6):442-447.
doi pubmed - Lian IA, Asberg A. Should total calcium be adjusted for albumin? A retrospective observational study of laboratory data from central Norway. BMJ Open. 2018;8(4):e017703.
doi pubmed - Pekar JD, Grzych G, Durand G, Haas J, Lionet A, Brousseau T, Glowacki F, et al. Calcium state estimation by total calcium: the evidence to end the never-ending story. Clin Chem Lab Med. 2020;58(2):222-231.
doi pubmed - Tee MC, Holmes DT, Wiseman SM. Ionized vs serum calcium in the diagnosis and management of primary hyperparathyroidism: which is superior? Am J Surg. 2013;205(5):591-596; discussion 596.
doi pubmed - Gauci C, Moranne O, Fouqueray B, de la Faille R, Maruani G, Haymann JP, Jacquot C, et al. Pitfalls of measuring total blood calcium in patients with CKD. J Am Soc Nephrol. 2008;19(8):1592-1598.
doi pubmed - Riancho JA, Arjona R, Sanz J, Olmos JM, Valle R, Barcelo JR, Gonzalez-Macias J. Is the routine measurement of ionized calcium worthwhile in patients with cancer? Postgrad Med J. 1991;67(786):350-353.
doi pubmed - Buege MJ, Do B, Lee HC, Weber DM, Horowitz SB, Feng L, Qing Y, et al. Corrected calcium versus ionized calcium measurements for identifying hypercalcemia in patients with multiple myeloma. Cancer Treat Res Commun. 2019;21:100159.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Endocrinology and Metabolism is published by Elmer Press Inc.
